A breakthrough in all-solid-state batteries has been achieved through the development of an eco-friendly solid electrolyte derived from Prussian Blue analogues (PBAs). This innovative research, conducted by a team of scientists from UNIST, promises to accelerate the commercialization of all-solid-state secondary batteries by addressing cost and environmental impact concerns.
Solid electrolytes play a crucial role in all-solid-state batteries, but traditional options have been hindered by high costs and environmental limitations. Led by Professor Hyun-Wook Lee and Professor Sung-Kyun Jung from the School of Energy and Chemical Engineering at UNIST, in collaboration with Professor Dong-Hwa Seo from the Korea Advanced Institute of Science and Technology (KAIST), the team utilized PBAs as a solid electrolyte, offering an economical and environmentally friendly alternative.
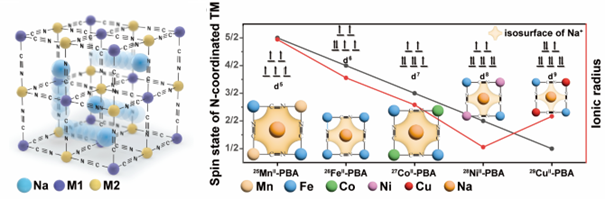
Figure 1. a) Schematic illustration of PBA framework and the Na+ diffusion path. b) Correlation between the spin state of N-coordinated transition metal ions and their lattice parameters.
Prussian Blue analogues (PBAs), historically known for their usage as blue dye materials since the 18th century, have emerged as a promising candidate for solid electrolytes due to their wide ion conduction channels and ease of synthesis. The research team discovered that the inherent properties of PBAs could enhance ionic conductivity, with different transition metals influencing the size of the ion channels and thus affecting conductivity.
Through comprehensive interfacial stability analysis with anode and cathode active materials, the research team identified suitable material groups and successfully developed an all-solid sodium secondary battery using manganese-based Prussian blue-based materials. The battery demonstrated a remarkable sodium ion conductivity of 0.1 mS/cm even at room temperature, showcasing the potential of PBAs as solid electrolytes.
The use of eco-friendly Prussian blue-based materials as solid electrolytes presents a new perspective for the future of solid electrolytes. This breakthrough research not only addresses the long-standing challenges of high prices and environmental concerns associated with existing solid electrolytes but also paves the way for the commercialization of all-solid-state batteries.
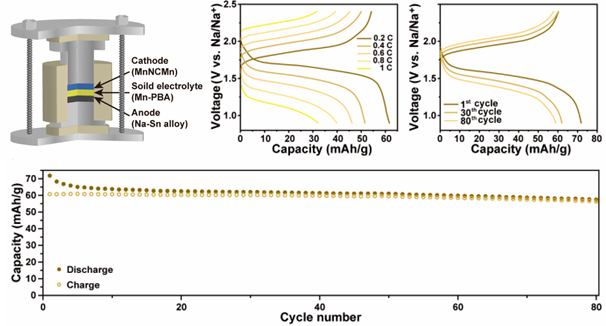
Figure 2. From top right are the schematic illustration of MnNCMn/SE/Na3Sn ASNBs using Mn-PBA as the SE, and the rate performance and charge–discharge voltage profile of the ASNBs at 30 °C as functions of the C-rates and number of cycles. On the bottom is the cycle performance of the ASNBs at a current density of 0.2C-rate and 30 °C.
Taewon Kim, the first author and a researcher in the School of Energy and Chemical Engineering, commented, “Through this study, we applied an eco-friendly Prussian blue-based material as a solid electrolyte. This study presents a new perspective on solid electrolytes for future applications.”
Sang Hyeok Ahn, another first author, added, “This study provides a solution to the persistent issues of high costs and environmental impact associated with existing solid electrolytes. It has the potential to greatly accelerate the commercialization of all-solid-state batteries in the future.”
Professor Hyun-Wook Lee emphasized the importance of expanding research possibilities beyond existing sulfide, oxide, and halide-based solid electrolytes, stating, “We hope that this discovery of new materials will broaden the possibilities in the field.” He further noted, “This study not only identifies these areas but also presents successful new solutions.”
This groundbreaking research has been published in the online version of the journal Angewandte Chemie International Edition on August 28, 2023. The study has received support from the 2023 Research Fund of UNIST, the KIST Institutional Program, and the Individual Basic Science & Engineering Research Program through the National Research Foundation (NRF) of Korea, funded by the Ministry of Science and ICT (MSIT).
Journal Reference
Taewon Kim, Sang Hyeok Ahn, You-Yeob Song, et al., “Prussian Blue-Type Sodium-ion Conducting Solid Electrolytes for All Solid-State Batteries,” Angew. Chem. Int. Ed., (2023).