A research team, affiliated with UNIST recently reported that the performance of electrodes for alkaline hydrogen evolution reaction (HER) can be significantly improve even without expensive electrocatalysts and complicated processes by modifying them with superaerophobic polymeric hydrogels. This breakthrough has been led by Professor Jungki Ryu and his research team in the Department of Energy Engineering at UNIST.
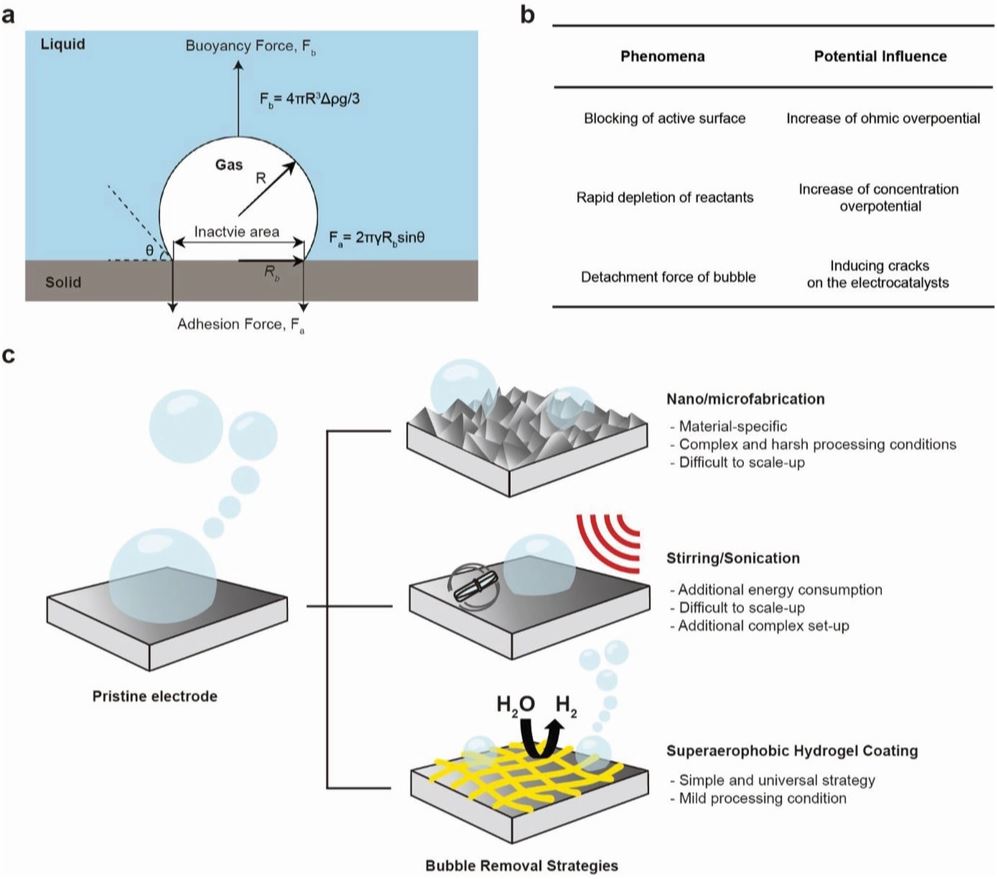
Figure 1. The scheme for a mechanism of bubble-detachment. a) Normal flat electrode, b) the problems of adhered bubbles, and c) various approaches for the removal of gas bubbles. In (a), R, ρ, g, and λ indicate the dimension of bubbles, the density of a solution, gravitational acceleration constant, and surface tension of a solution, respectively.
In this study, the research team reported a simple strategy to enhance the efficiency of electrochemical hydrogen production by imparting superaerophobicity to an underlying electrode with porous polymeric hydrogels. Superaerophobic hydrogels were readily coated on target substrates by cross-linking polyethyleneimine (PEI) via Schiff-base condensation reactions followed by freeze-drying, noted the research team. As a result, they could readily control the pore size, porosity, and superaerophobicity of the hydrogel-coated electrodes by varying the concentrations of PEI upon cross-linking. Due to facile removal of as-generated hydrogen bubbles, the NF electrode modified with PEI hydrogel only outperformed those modified with expensive electrocatalysts especially at high current densities, according to the research team.
“We believe that our results can pave the way for the practical application of water electrolysis by providing insights into the design of electrodes and electrolyzers,” noted the research team.

Figure 2. Their findings have also been featured as a front cover of Advanced Energy Materials on August 4, 2022.
This study has been participated by Misol Bae in the Department of Energy Engineering at UNIST. Published in the August 2022 issue of Advanced Energy Materials, their findings have also been featured as a front cover.
Journal Reference
Misol Bae,Yunseok Kang,Dong Woog Lee, et al., “Superaerophobic Polyethyleneimine Hydrogels for Improving Electrochemical Hydrogen Production by Promoting Bubble Detachment,” Adv. Energy Mater., (2022).