A research team, led by Professor Yongwon Seo in the Graduate School of Carbon Neutrality at UNIST has unveiled a highly efficient method for desalinating seawater using hydrate-based desalination (HBD) technology. The breakthrough is expected to have far-reaching implications for the application of hydrate-based desalination techniques, with the ability to calculate optimal temperatures for enhanced efficiency.
Hydrate desalination technology, known for its eco-friendly freshwater production capabilities, offers a low-energy solution that can be effectively used in treating high concentrations of brine or contaminated water. By leveraging the phenomenon where impurities, including salts, are expelled during the formation of hydrates from brine or contaminated water, clean water can be obtained.
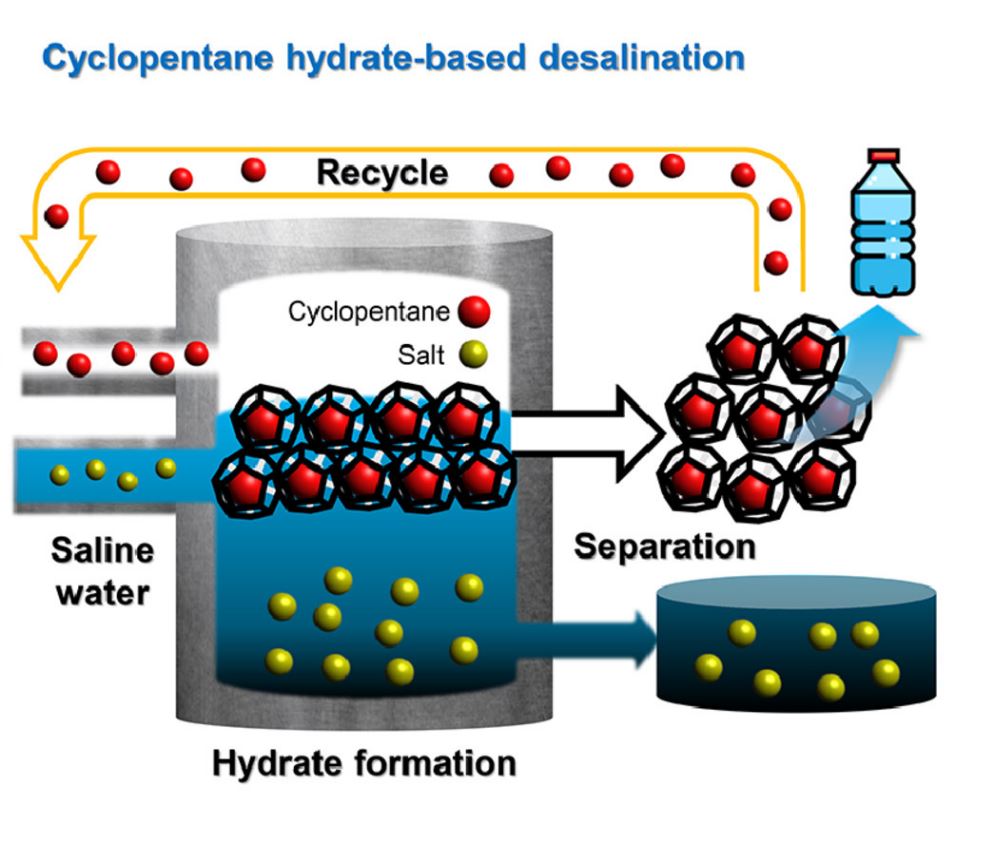
Figure 1. Schematic image, showing the cyclopentane (CP) HBD.
In this study, the research team experimentally measured the thermodynamic properties of cyclopentane hydrate and evaluated the efficiency of the hydrate desalination technique based on these measurements. They also proposed a novel calculation method for predicting the temperature that maximizes desalination efficiency.
“When specific conditions are met after adding cyclopentane, a colorless volatile liquid, to brine, cyclopentane hydrate, consisting of pure water, is formed,” explained Professor Seo. “We experimentally measured the thermodynamic properties of this cyclopentane hydrate to apply it to desalination processes.”
Building upon their experiments, the research team calculated the maximum water yield of cyclopentane hydrate under various conditions. The efficiency was determined based on the maximum water yield and the thermal energy required for cooling during the hydrate desalination process. A thermodynamic relational equation was developed to identify the temperature at which maximum efficiency can be achieved for each brine concentration.
For instance, when the hydrate desalination technique is applied to brine with initial salt concentrations of 3.5 wt% and 5 wt%, the relational equation calculates the energy efficiency optimum temperatures as 273.4 K and 271.5 K, respectively. The corresponding maximum water yields are 67% and 61.1%. This research provides a crucial criterion for optimizing the efficiency of real-world desalination processes.
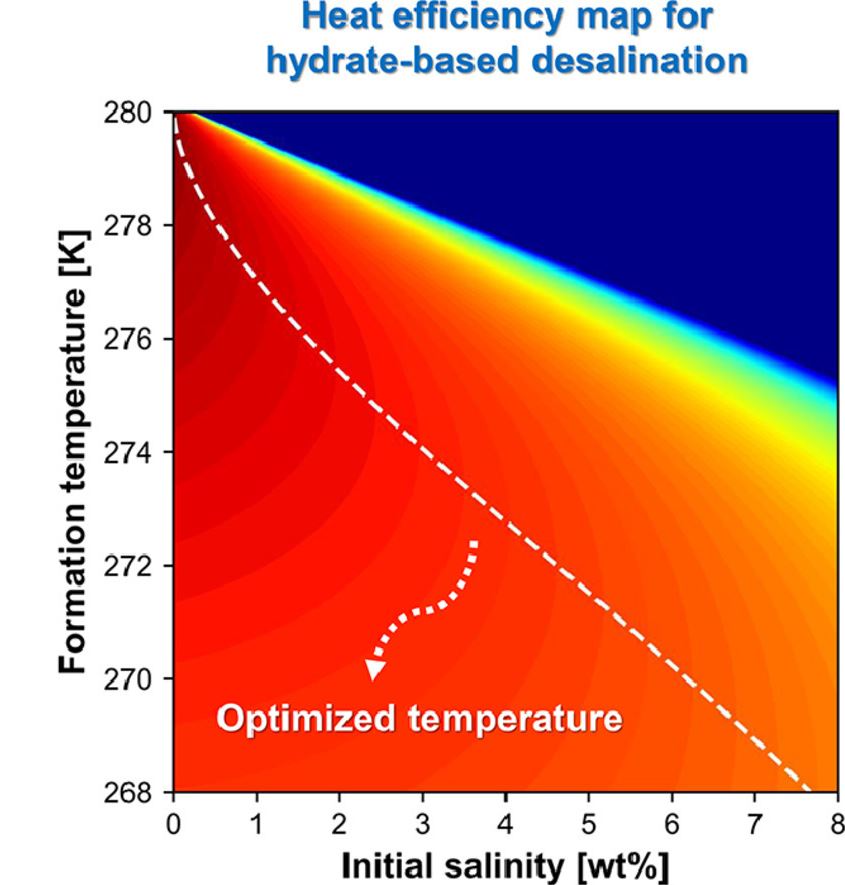
Figure 2. Schematic image, showing the heat efficiency map for HBD.
Junghoon Mok, the first author of the study, emphasized the significance of this research, stating, “This study is expected to optimize the energy consumption in high-concentration brine treatment processes using hydrate freshwater techniques.” He added, “The proposed approach is not only applicable to hydrate-based desalination but also holds promise for freeze-type desalination technology.”
The study was conducted in collaboration with the Korea Institute of Production and Technology and received support from the Ministry of Science and ICT’s Korea Research Foundation’s basic research project, nuclear research and development project, and the Korea Energy Technology Evaluation Institute.
This research, conducted in collaboration with the Korea Institute of Industrial Technology (KITECH), has received support from the Korea Institute of Energy Technology Evaluation and Planning (KETEP), the Ministry of Trade, Industry and Energy (MOTIE), and the National Research Foundation of Korea (NRF). The paper, detailing this groundbreaking research, was published in the November 2023 issue of Water Research.
Journal Reference
Junghoon Mok, Minseo Park, Wonjung Choi, et al., “Investigation of theoretical maximum water yield and efficiency-optimized temperature for cyclopentane hydrate-based desalination,” Water Res., (2023).












