It is well known that cancerous tumor cells have an acidic pH microenvironment (pH 5.6 to 6.8). Using this unique feature, researchers have developed a new anticancer therapeutic agent that selectively kills cancer cells. This access allows detached malignant cells from the tumor to penetrate into cancer cells and induce mitochondrial dysfunction, thereby killing only cancer cells.
This breakthrough has been developed by Professor Ja-Hyoung Ryu and his research team in the Department of Chemistry at UNIST.
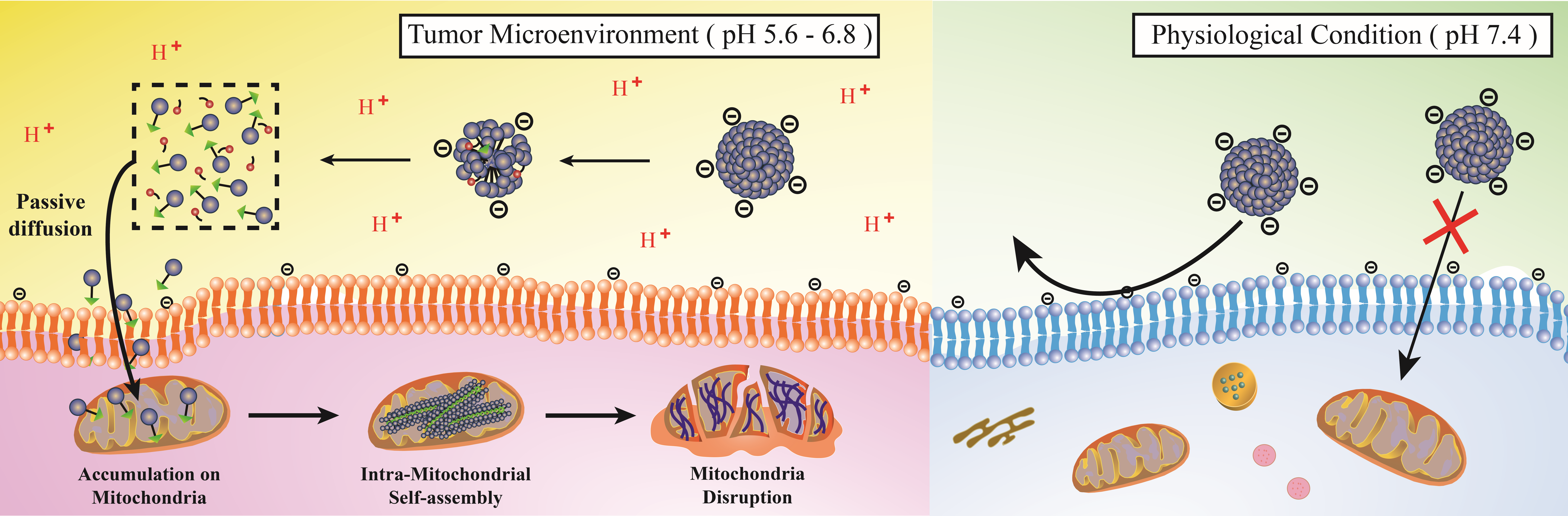
Figure 1. Schematic illustration of disassembly–assembly approach of mitochondria targeted conventional drug free agents with enhanced selectivity. Chemical structure of pH responsive molecule Mito-SA, consisting of pH responsive succinic anhydride moiety. The molecules are stable under physiological conditions and prohibit the entry inside the normal cell. In contrast, stimulated by the acidic pH, the Mito-SA micelle disassemble near the cancer cell to the parental molecule Mito-FF which then diffuse to the cell, target mitochondria, induce fiber to destroy mitochondria leading to apoptosis.
In the study, the research team developed transformable nano-assemblies of a mitochondria-targeting agent, Mito-SA, to achieve enhanced tumor selectivity. According to the research team, the new substance formed micelles containing a negative surface charge that selectively disassembled near the tumor cells into parental positively charged Mito-FF molecules, which induced apoptosis via self-assembly into nanofibers in the cancer cell mitochondria.
However, the entry of the Mito-SA micelle inside the normal cell is restricted due to the repulsion between the negatively charged micelle and the negatively charged plasma cell membrane, noted the research team. Furthermore, their findings also revealed that Mito-SA displayed an ideal tumor reduction ability and showed no side effect/damage to the normal tissue.
“Our work shows a strategy for the efficient delivery of positively charged, mitochondria-targeting agents by developing charge-shielded nano-assemblies that selectively disassemble in the tumoral environment,” said the research team.
Their findings have been published in the December 2022 issue of Advanced Functional Materials. This study has been carried out in collaboration with Professor Sang Kyu Kwak from Korea University and Professor Sehoon Kim from the KU-KIST Graduate School of Converging Science and Technology.
Journal Reference
M. T. Jeena, Seongeon Jin, Keunsoo Jeong, et al., “Cancer-Selective Supramolecular Chemotherapy by Disassembly-Assembly Approach,” Adv. Funct. Mater., (2022).












