Revolutionary vascular progenitor cells (VPCs) for the treatment of ischemic vascular diseases has been discovered. This has been generated by direct lineage conversion, which is less likely to cause cancer during large-scale cell culture, and thus can be used as the main raw material for blood vessels needed for 3D printing of human tissues, such as heart or liver.
This breakthrough has been led by Professor Jeong Beom Kim and his research team in the School of Life Sciences at UNIST. In the study, the research team demonstrated that induced VPCs (iVPCs) can be generated from fibroblasts by ETS (E-twenty six) transcription factors, Etv2 and Fli1. In addition, transplantation of this iVPCs into the ischemic hindlimb model enhanced blood flow without tumor formation in vivo, which offers intriguing possibilities as therapeutic agents for treating ischemic vascular diseases.
When damage occurs to blood vessels that transport blood cells, nutrients, and oxygen to the tissues of the body, ischemic vascular disease occurs. Cell therapy that involves transplanting cells to restore tissue or organ function has received much attention, as a potential treatment for ischemic diseases. Vascular progenitor cells (VPCs), which were generated through the differentiation of pluripotent stem cells (PSCs), including embryonic stem cells and induced PSCs (iPSCs), have self-renewal capacity, and thus are well-suited for mass production. Although VPCs are deemed the strongest candidate for treatment of ischemic diseases, the residual undifferentiated cells could form tumor after transplantation, a critical obstacle for the application of PSC-derived VPCs to ischemic disease patients.
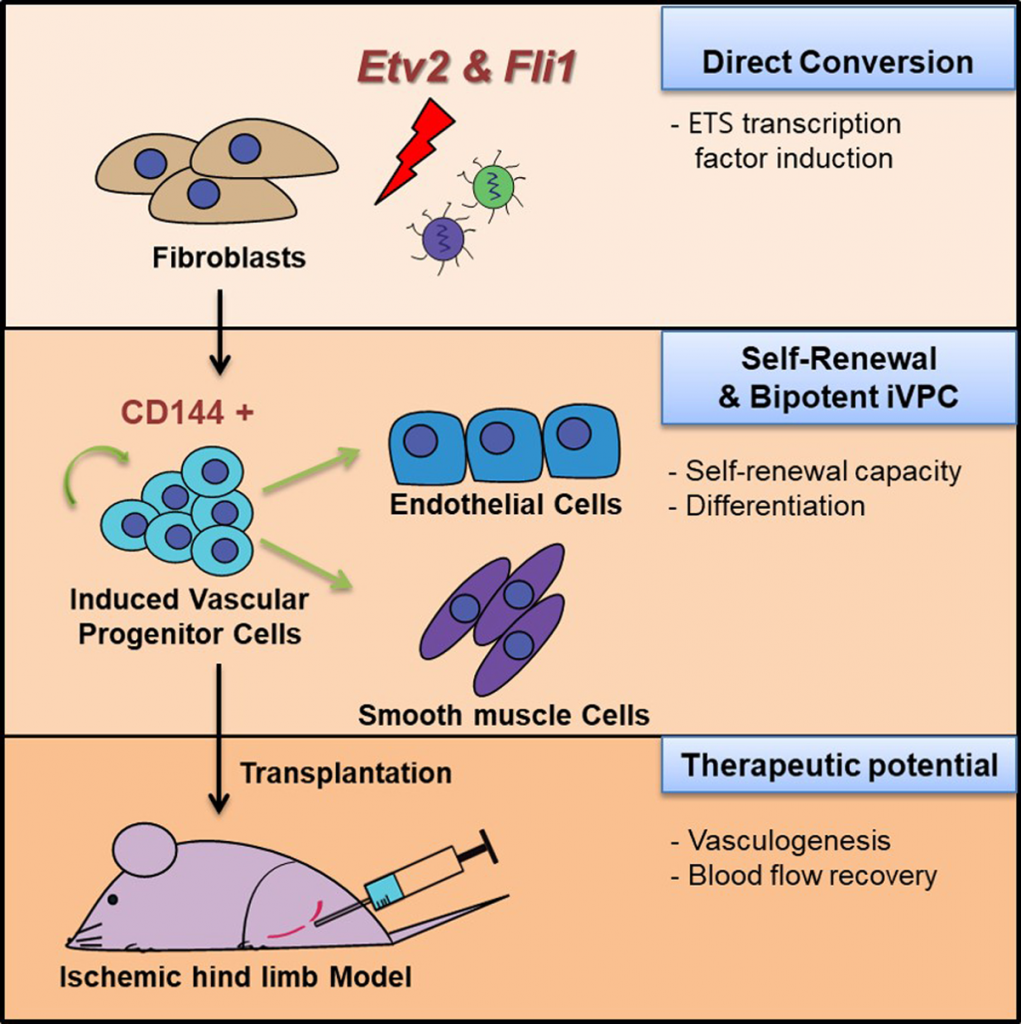
Professor Kim has solved this issue by direct lineage conversion, the induction of functional cells from another lineage by using only lineage-restricted transcription factors. In the study, the research team demonstrated that induced VPCs (iVPCs) can be generated from fibroblasts by ETS (E-twenty six) transcription factors, Etv2 and Fli1.
“Both DNAs of Etv2 and Fli1 are mainly expressed in the early stages of vascular development,” says Soo Yong Park (Combined M.S/Ph.D. of Energy and Chemical Engineering, UNIST), the first author of the study. “In this study, we demonstrate that the ectopic expression of Etv2 and Fli1 are sufficient to convert fibroblasts into induced VPCs (iVPCs) that can give rise to both ECs and SMCs.”
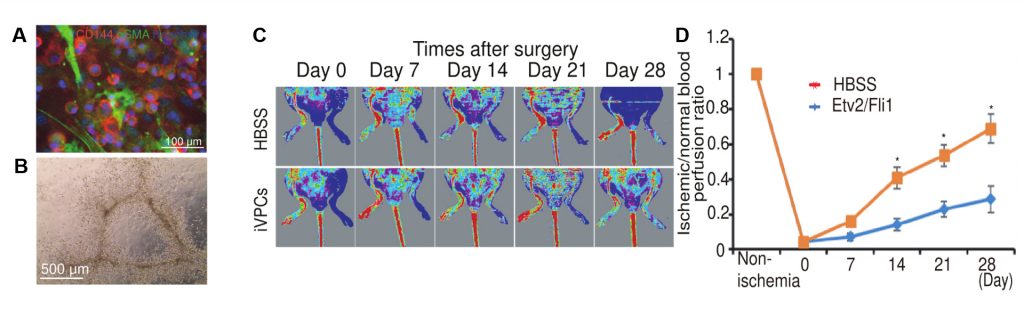 Stem cells have the capacity to proliferate extensively (self-renewal) and differentiate into other cell types (biopotent). In the study, the research team has successfully generated bipotential self-renewing iVPCs by direct lineage conversion. They also confirmed that iVPCs could differentiate into functional endothelial cells (iVPC-derived endothelial cells) or smooth muscle cells (iVPC-derived smooth muscle cells) in defined culture conditions. Besides, they observed that transplanted iVPCs enhanced blood flow without tumor formation in an ischemic vascular disease model.
Stem cells have the capacity to proliferate extensively (self-renewal) and differentiate into other cell types (biopotent). In the study, the research team has successfully generated bipotential self-renewing iVPCs by direct lineage conversion. They also confirmed that iVPCs could differentiate into functional endothelial cells (iVPC-derived endothelial cells) or smooth muscle cells (iVPC-derived smooth muscle cells) in defined culture conditions. Besides, they observed that transplanted iVPCs enhanced blood flow without tumor formation in an ischemic vascular disease model.
“We demonstrated that ischemic disease curable iVPCs, which have self-renewal and bipotency, can be generated from mouse fibroblasts by enforced ETS family transcription factors, Etv2 and Fli1 expression,” says Professor Kim. “Our study may facilitate future therapeutic application for atherosclerosis.”
This study has been jointly carried out with Professor Kim’s startup company, SuPine Therapeutics Inc. with the support of the Ministry of SMEs and Startups (MSS). The findings of this reseaarch have been published in the journal, Arteriosclerosis, Thrombosis, and Vascular Biology on March 25, 2020.
Journal Reference
Soo Yong Park, Hyunah Lee, Yang Woo Kwon, et al., “Etv2- and Fli1-Induced Vascular Progenitor Cells Enhance Functional Recovery in Ischemic Vascular Disease Model—Brief Report,” ATVB (2020)












