A groundbreaking discovery has been made by Professor Hyug Moo Kwon and his research team in the Department of Biological Sciences at UNIST, in collaboration with Professor Jaeseok Yang from Yonsei University. Their study sheds new light on the protein called ‘TonEBP,’ revealing its significant role in the development of lupus and lupus nephritis. This breakthrough not only enhances our understanding of these conditions, but also opens up potential avenues for future treatment options.
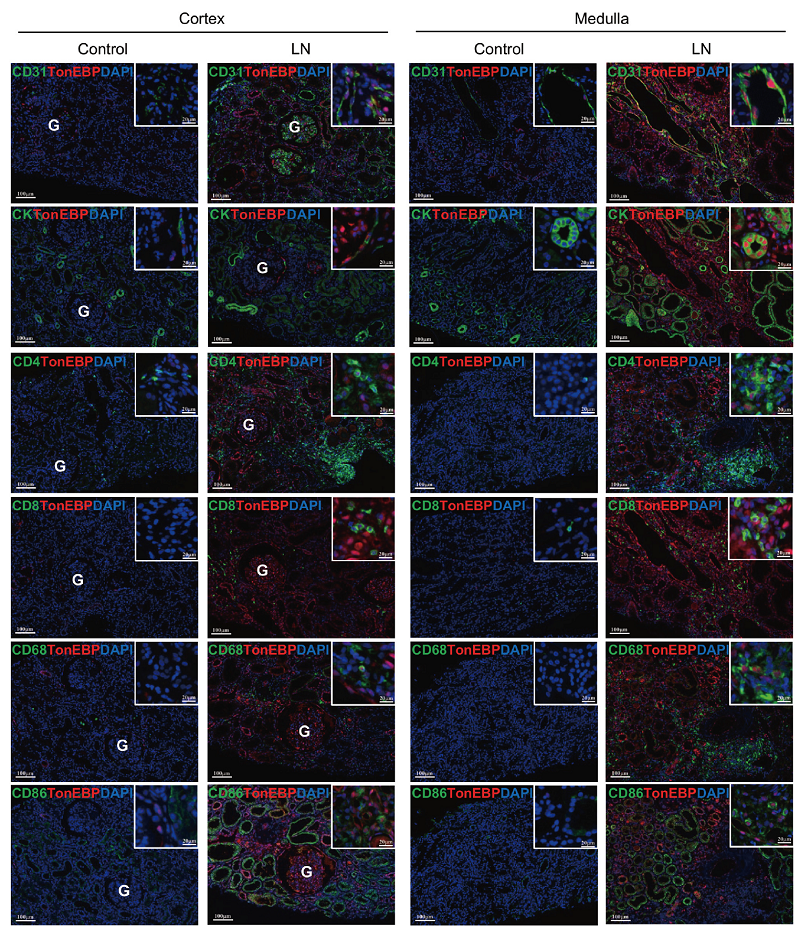
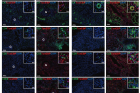
Figure 1. TonEBP expression in endothelial cells, epithelial cells, and infiltrating immune cells in renal biopsy tissues subjected to multiplex immune fluorescence staining. Biopsy sections were double stained for TonEBP (red) and cell surface markers (green): CD31, cytokeratin (CK), CD4, CD8, CD68, or CD86. Nuclei were visualized with 4′,6-diamidino-2-phenylindole (DAPI; blue). Representative pseudocolor merged images of the renal cortex and medulla are shown for both controls and the patients with lupus nephritis (LN). Merged image demonstrates colocalization of TonEBP and DAPI in the nucleus (purple) and cell markers on the cell surface and cytoplasm (green). Original magnification ×200 (bars = 100 μm) and ×400 (boxed areas; bars = 20 μm). G, glomerulus.
Lupus is an autoimmune disorder characterized by autoreactive B cells and dysregulation of various immune cells, including myeloid cells. Lupus nephritis (LN), which refers to kidney inflammation caused by lupus, is one of the most common organ manifestations associated with systemic lupus erythematosus (SLE). The study conducted meticulous analysis on kidney biopsy samples obtained from LN patients to investigate TonEBP’s involvement in these conditions.
The findings unveiled elevated expression levels of TonEBP not only within kidney cells but also infiltrating immune cells when compared to control patients. This increased expression correlated with inflammatory cytokines involved in inflammatory reactions triggered by bacterial or viral infections, as well as severity of proteinuria—a key indicator reflecting kidney damage.
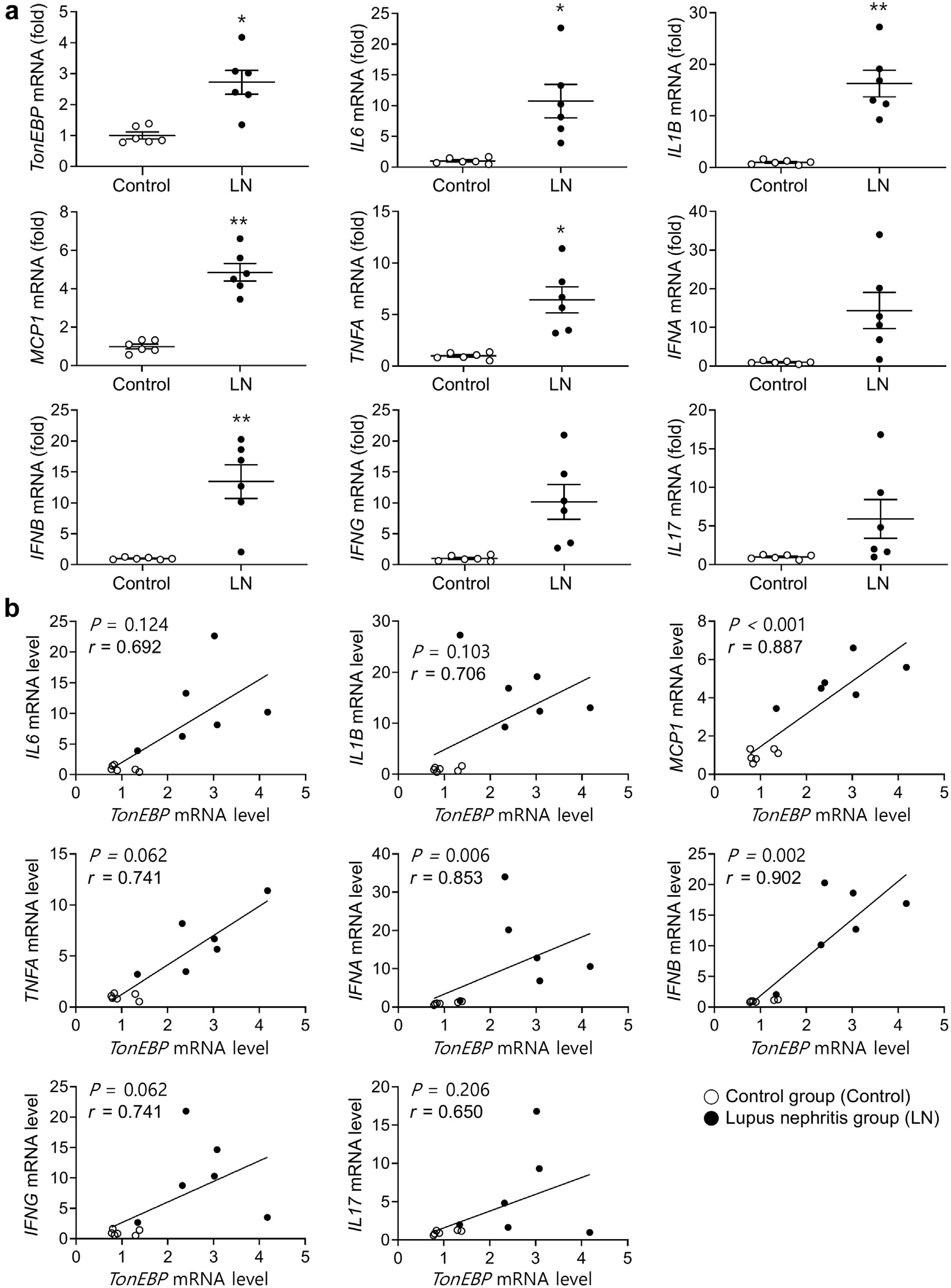
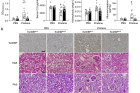
Figure 2. Renal expression of mRNA encoding TonEBP and proinflammatory cytokines in patients with lupus nephritis (LN). (a) Levels of mRNA encoding TonEBP and proinflammatory cytokines were measured by quantitative real-time polymerase chain reaction of frozen kidney biopsy samples from patients with LN and controls (Table 1). mRNA level of each gene was normalized to that of glyceraldehyde-3-phosphate dehydrogenase. Mean ± SEM. ∗P < 0.05, ∗∗P < 0.01 compared with the controls. (b) Correlation between expression of each cytokine mRNA and that of TonEBP.
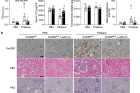
To delve deeper into TonEBP’s impact on both lupus development and kidney damage, animal experiments were conducted. The results demonstrated that specifically suppressing TonEBP within myeloid cells effectively halted lupus progression while mitigating kidney damage. Additionally, animal models with myeloid deficiency of TonEBP exhibited elevated efferocytosis (phagocytic clearance) alongside reduced differentiation of Th1 (T helper 1) and Th17 (T helper 17) cells—indicating impaired Toll-like receptor (TLR) signaling, which is crucial for T cell function.
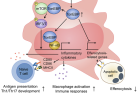
Furthermore, the study revealed that various TLRs stimulated macrophages to induce TonEBP expression through promoter activation—a critical intracellular signaling pathway dependent on TonEBP. This finding suggests that TonEBP acts as a transcriptional cofactor for NF-κB and activates mTOR-IRF3/7 via protein-protein interactions.
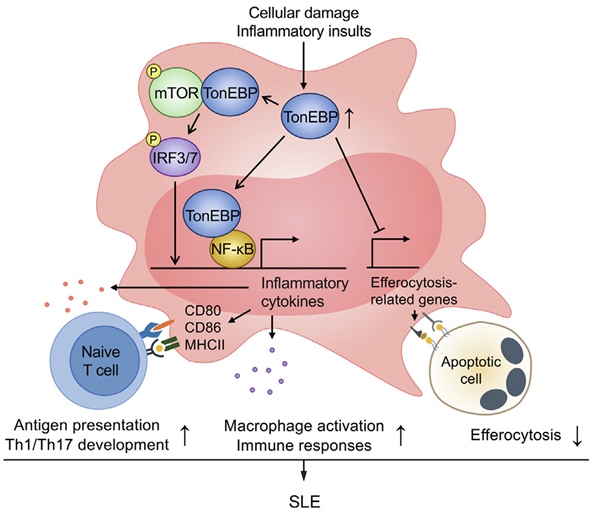
Figure 3. Proposed model for the role of macrophage TonEBP in systemic lupus erythematosus (SLE).
“This study unveils the pivotal role of TonEBP in lupus development,” stated Professor Kwon. “By identifying the underlying cause and providing valuable clues for new treatments, we aim to contribute to improving the lives of individuals affected by these conditions.”
The research findings were made available on May 5 ahead of their official publication in Kidney International on July 1, 2023. The study received support from grants provided by the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT (MSIT).
Journal Reference
Jun Kyu Choe, Junsoo Kim, Hyeonseo Song, et al., “Macrophage transcription factor TonEBP promotes systemic lupus erythematosus and kidney injury via damage-induced signaling pathways,” Kidney Int., (2023).