Professor Chan Young Park and his research team from the Department of Biological Sciences at UNIST have discovered that cilia damage in infected cells occurs due to the interaction between the main protease (MPro), a key protein in the virus’ life cycle and septin proteins that regulate cell division, movement, and maintain cell structure.
Cilia are essential for various life activities, such as embryonic development, maintaining homeostasis, preventing pathogens from entering respiratory tracts, and transmitting signals from olfactory nerve cells. The Sonic Hedgehog (SHH) signaling pathway is a representative method of ciliary-specific signaling involved in tissue regeneration, generation, cell differentiation, and division.
Recent studies have revealed that coronavirus infections cause significant damage to respiratory cilia. However, details regarding the mechanism behind this phenomenon or abnormalities in ciliary signal pathways are not yet well understood.
Coronaviruses, such as the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) responsible for causing the COVID-19 pandemic, infect human cilia and utilize host cell resources to produce virus genes and proteins. The replication of coronaviruses requires protein cleavage (protease), which cuts the protein chain produced by the virus into various functional proteins necessary for their propagation.
However, recent research indicates that coronavirus proteases not only cut virus-specific proteins but also those present in infected cells resulting in viral infection or damage.
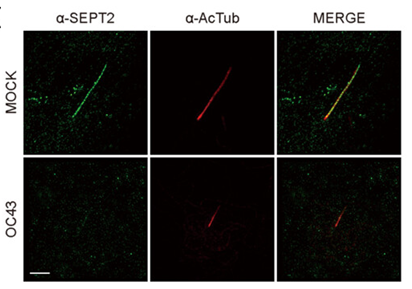
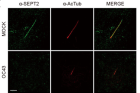
Figure 1, Shown above, illustrates how HCoV-OC43 infection reduces SEPT2 and increases deciliation in ciliary cells. The figure displays superresolution microscopy of SEPT2 localization along with the axoneme and reduced localization due to OC43 infection. The image shows SEPT2 (in green) and acetylated α-tubulin (in red), with a scale bar of 2 µm.
In this study, the research team demonstrated that coronavirus infection and the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) 3CL protease induce cilia dysfunction by targeting a host protein called septin, which is essential for maintaining the structure and function of cilia.
They also found that coronaviruses and 3CL protease (3CLpro) cause cleavage of several septin proteins (SEPT2, -6, and -9), producing obstructive fragments that result in defective ciliogenesis, disoriented septin filaments, and ablated SHH signaling. Furthermore, their study shows that ectopic expression of cleaved SEPT2 fragments leads to abnormal ciliary structures with dysfunctional SHH signaling in a protease activity-dependent manner. According to the research team, their findings suggest that inhibitors of 3CLpro are potent in preventing abnormal ciliary structures along with SHH signaling.
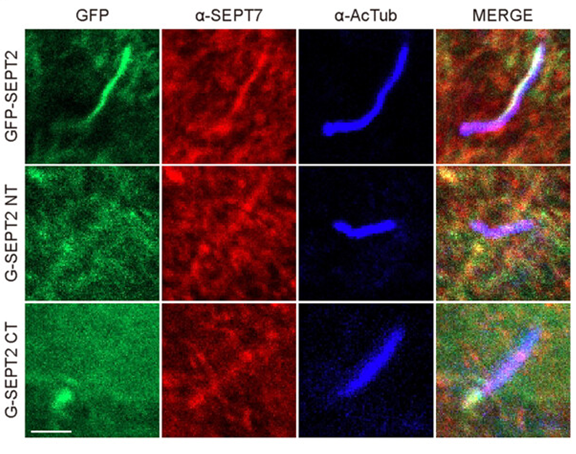
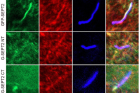
Figure 2. The image above demonstrates how cleaved SEPT2 fragments affect ciliogenesis by preventing the translocation of ciliary SEPTs. Specifically, hTERT-RPE1 cells expressing GFP-SEPT2 fragments (FL, NT, and CT) were serum-starved for 24 hours and immunostained with acetylated α-tubulin (in blue) along with SEPT7 (in red; panel G) or SEPT9 (in red; panel H). The scale bar in the image is 2 µm.
“These results provide valuable insights into understanding the general mechanisms underlying ciliary defects caused by coronaviruses,” noted the research team. “Moreover, they highlight how preclinical as well as clinical trials on 3CL protease inhibitors could be useful therapeutic options for treating virus-induced defects associated with cilium malfunctioning.”
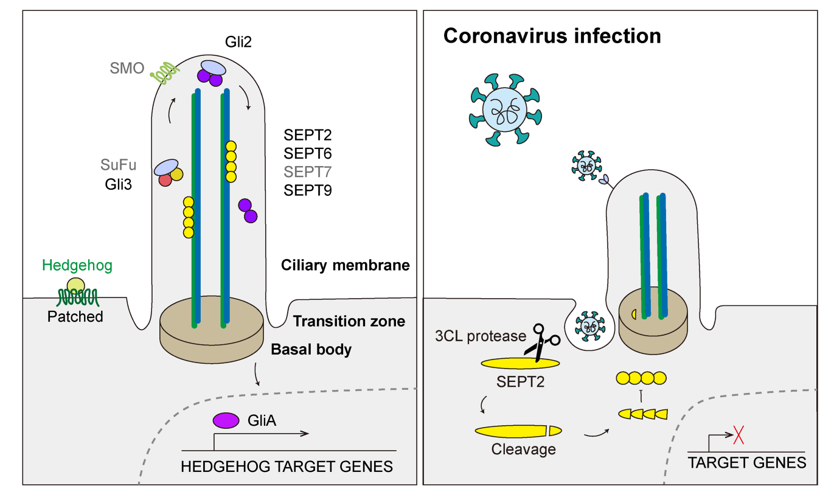
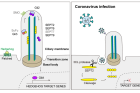
Figure 3, Illustrated above, shows how coronavirus infection induces cilia damage and suppresses intracellular signaling mechanisms.
The study findings have been published ahead of their official publication in the online version of the Journal of Medical Virology on February 24, 2023. This study received support from the National Research Foundation of Korea (NRF) and Ministry of Science and ICT (MSIT) through its Global Ph.D. Fellowship and Mid-Career Researcher Programs.
Journal Reference
Ah Reum Lee, Yeong Cheon Kweon, Sang Min Lee, Chan Young Park, “Human coronavirus 3CL proteases cleave septins and disrupt Hedgehog signaling, causing ciliary dysfunction,” J. Med. Virol. (2023).