A novel porous material capable of separating deuterium (D2) from hydrogen (H2) at a temperature of 120 K (-153°C) has been introduced. Notably, this temperature exceeds the liquefaction point of natural gas, which is 111 K (-162°C), by more than 10°C, thus facilitating large-scale industrial applications. This advancement presents an attractive pathway for the economical production of D2 by leveraging the existing infrastructure of liquefied natural gas (LNG) production pipelines.
The research team, jointly led by Professor Hyunchul Oh from the Department of Chemistry at UNIST, Professor Jaheon Kim from Soongsil University, Dr. Jitae Park from Heinz Maier Leibnitz Zentrum (MLZ) at Technical University of Munich (TUM), and Dr. Margarita Russina from Helmholtz-Zentrum Berlin für Materialien und Energie (HZB) in Berlin, Germany announced this advancement on March 19, 2025. The study also involved Minji Jung, Jaewoo Park, and Raeesh Muhammad from the Department of Chemistry at UNIST, who served as co-first authors.
Deuterium, a stable isotope of hydrogen, plays a critical role in enhancing the durability and luminous efficiency of semiconductors and display devices, as well as serving as a fusion fuel in energy production. However, the increasing demand for D2 presents challenges in its production, primarily due to the need to separate from hydrogen through a cryogenic distillation process conducted at temperatures as low as 20 K (-253°C). While research has explored the use of metal-organic frameworks (MOFs) for D2 separation, their efficiency diminishes significantly at elevated temperatures.
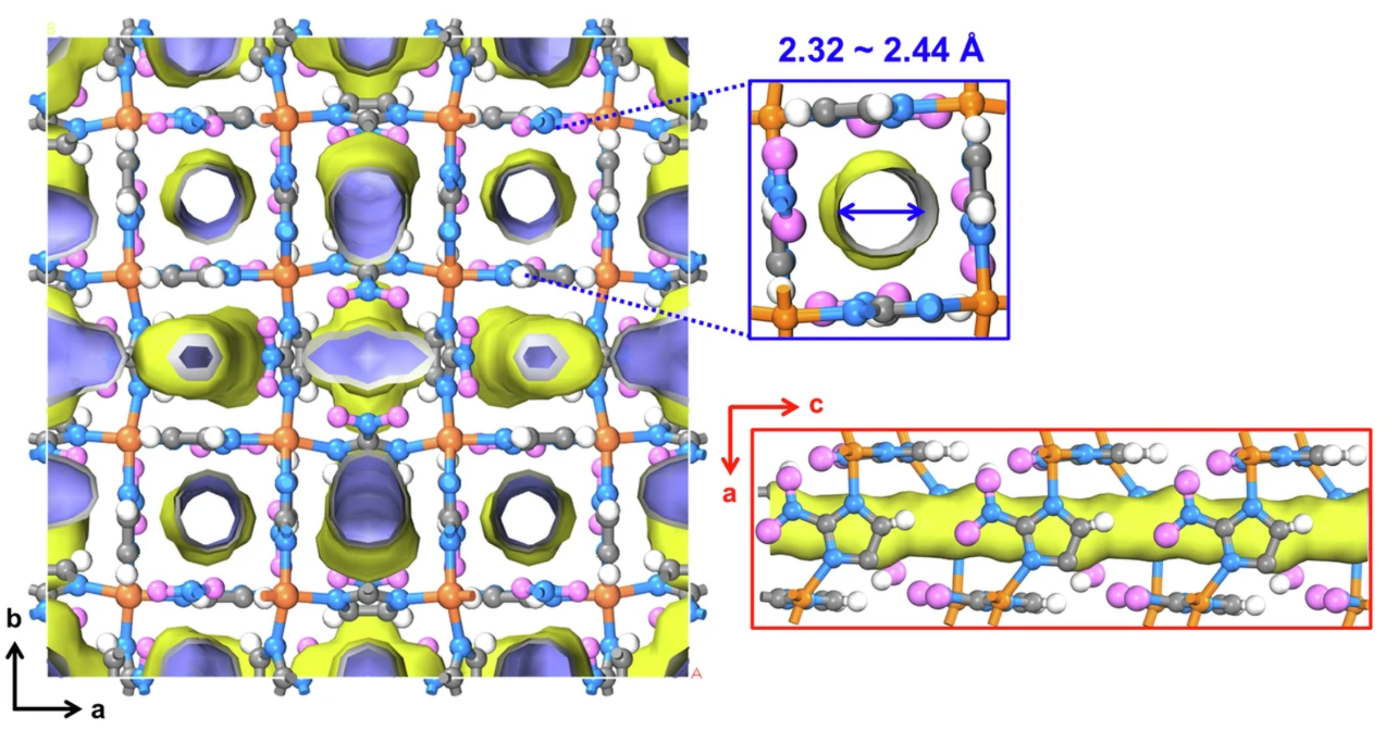 Figure 1. The crystal structure of Cu-ZIF-gis that shows cylindrical straight channels along the c-axis31. The pores were calculated with Connolly surfaces with a probe of 1.1 Å. (Cu, orange; N, blue; C, gray; O, magenta; H, white).
Figure 1. The crystal structure of Cu-ZIF-gis that shows cylindrical straight channels along the c-axis31. The pores were calculated with Connolly surfaces with a probe of 1.1 Å. (Cu, orange; N, blue; C, gray; O, magenta; H, white).
In this study, the research team presented a copper-based zeolite imidazolate framework (Cu-ZIF-gis), which shows exceptional D2 separation performance, even at 120 K (-153℃). While typical MOFs operate effectively at around 23 K (-250℃), their performance decreases sharply as temperatures approach 77 K (-196℃). However, the newly developed Cu-based MOF demonstrates a significant advantage in maintaining its effectiveness at higher temperatures.
For the first time, the research team identified that the superior performance of this material results from the increased expansion of its lattice as the temperature rises. At cryogenic temperatures, the pores of the developed MOF are smaller than H2 molecules, thereby inhibiting their passage. However, as the temperature increases, the lattice expands, leading to an increase in pore size. This enlargement facilitates the passage of gases through the pores, thereby enabling the separation of H2 and D2 via the quantum sieving effect, wherein heavier molecules traverse the pores more efficiently at lower temperatures.
Confirmatory in-situ X-ray diffraction (XRD) and quasi-elastic neutron scattering (QENS) experiments, conducted by the joint team from UNIST, HZB, and MLZ at the Institut Laue-Langevin (ILL) in Grenoble, France, confirmed the expansion of the lattice framework with increasing temperature, as well as the difference in isotope diffusivity even at elevated temperatures. Additionally, the analysis from the Thermal Desorption Spectroscopy (TDS) experiments indicated stable D2 separation at elevated temperatures.
Professor Oh remarked, “The reported material exhibits markedly lower energy consumption and enhanced separation efficiency compared to most traditional methods, which operate at extremely low temperatures.” Dr. Jitae Park further noted, “These findings can be applied to develop sustainable isotope separation technologies using existing LNG cryogenic infrastructure, underscoring its potential industrial impact.”
Dr. Margarita Russina highlighted the crucial role of QENS in this study, stating: “With QENS, we can directly probe the molecular motion of H2 and D2 in MOFs, gaining key insights into their diffusion behavior and interactions with porous materials. The observed stronger confinement of D2 compared to H₂, a strictly nanoscale phenomenon, leads to remarkable effects on macroscopic properties, forming the basis for the development of a new generation of materials for more efficient isotope separation.”
The findings of this research have been published in Nature Communications on February 27, 2025. This study was supported by the National Research Foundation (NRF) of Korea and the Ministry of Science and ICT (MSIT), and the Institut Laue-Langevin (ILL) in Grenoble, France for the allocation of beam time.
Journal Reference
Minji Jung, Jaewoo Park, Raeesh Muhammad, Taeung Park, Sung-Yeop Jung, Jungwon Yi, Cheolwon Jung, Jacques Ollivier, Anibal J. Ramirez-Cuesta, Jitae T. Park, Jaheon Kim, Margarita Russina, and Hyunchul Oh, “Lattice-driven gating in a Cu-based zeolitic imidazolate framework for efficient high-temperature hydrogen isotope separation,” Nat. Commun., e202417137 (2025).