A team of researchers, affiliated with UNIST have unraveled the molecular mechanisms behind the activation of multiple inflammasomes and their role in inducing inflammatory cell death. This discovery sheds light on the connection between innate immune sensors and provides valuable insights into the functions of inflammasomes in innate immunity and inflammasome biology. The findings also hold potential for identifying new therapeutic targets for diseases mediated by NLRP3, AIM2, NLRC4, and Pyrin.
Inflammasomes serve as crucial sentinels of innate immune defense, sensing pathogens and triggering cell death in infected cells, thereby playing vital roles in inflammation, development, and cancer. These inflammasome sensors detect specific pathogen- and damage-associated molecular patterns (PAMPs and DAMPs) and form a complex with the adapters ASC and caspase-1. However, the integration of multiple inflammasome sensors for optimal host defense has remained elusive.
The research team, led by Professor SangJoon Lee from the Department of Biological Sciences at UNIST, investigated the simultaneous activation of multiple inflammasomes by various ligands. They discovered that this activation triggered distinct types of programmed inflammatory cell death, which could not be replicated by treating with a single pure ligand of any individual inflammasome. The team identified NLRP3, AIM2, NLRC4, and Pyrin as members of a large multiprotein complex, along with ASC, caspase-1, caspase-8, and RIPK3. This complex was found to drive a process called PANoptosis, involving multiple types of inflammatory cell death.
Furthermore, the multiprotein complex was released into the extracellular space, forming multiple extracellular inflammasome particles. These particles, when engulfed by neighboring macrophages, induced inflammation. This discovery highlights a previously unknown regulatory connection and molecular interaction between inflammasome sensors, leading to the assembly of a multiprotein complex that includes multiple inflammasome sensors and cell death regulators.
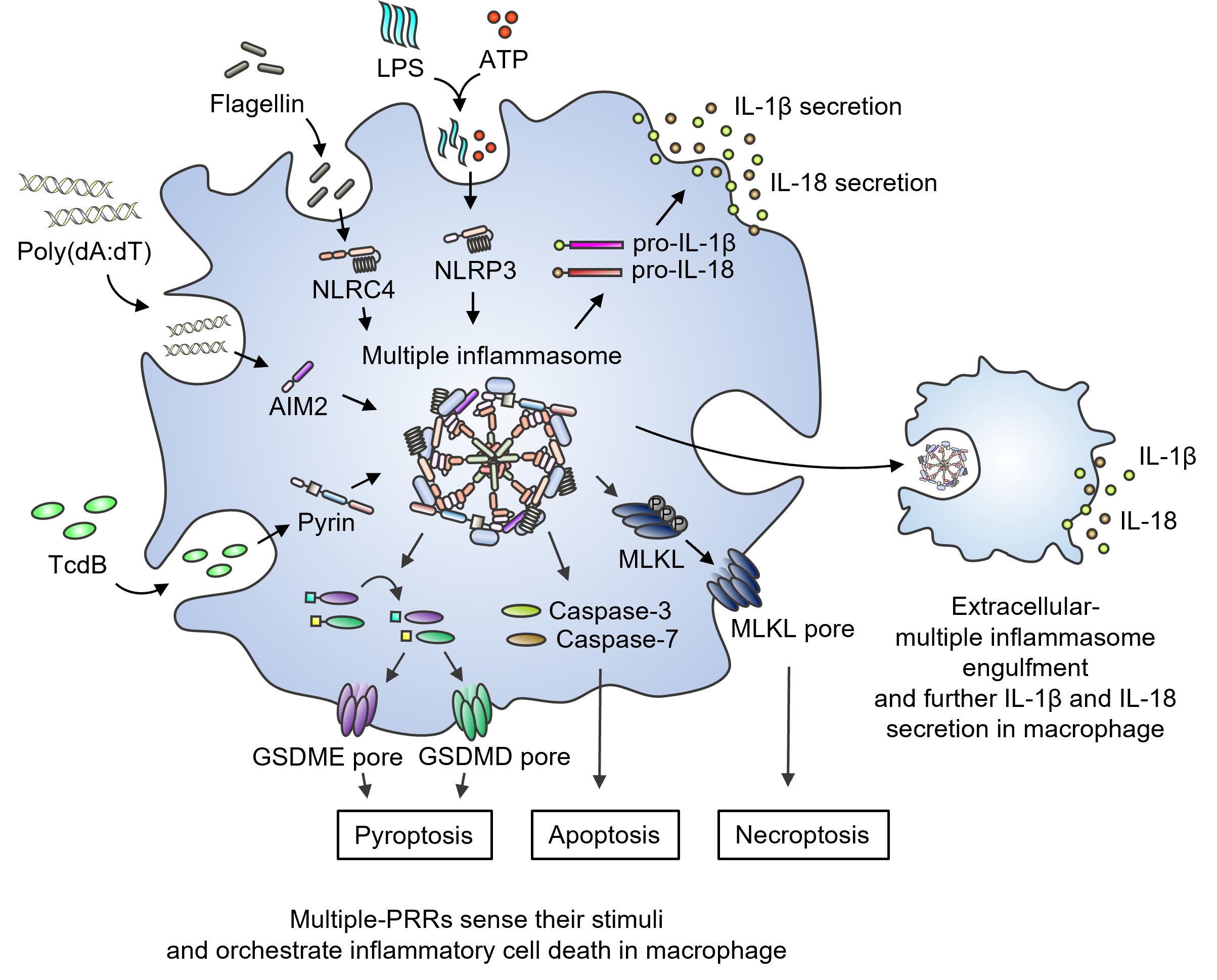
Figure 1. The Formation of Panoptosomes and Inflammatory Apoptosis Following the Injection of Four Pathogen-Related Patterns
The implications of these findings extend beyond fundamental research. Understanding the critical interactions among NLRP3, AIM2, NLRC4, and Pyrin opens up new avenues for comprehending the functions of these molecules in innate immunity and inflammasome biology. Moreover, it paves the way for identifying potential therapeutic targets for diseases mediated by these inflammasomes.
Professor Lee emphasized the significance of this research, stating, “Our study provides a new paradigm for understanding the functions of inflammasome sensors and their impact on innate immunity. The identification of key molecular interactions and the assembly of the multiprotein complex shed light on the intricate mechanisms of immune defense against multiple pathogens. This knowledge can potentially guide the development of novel therapeutic strategies for diseases associated with NLRP3, AIM2, NLRC4, and Pyrin.”
The research team conducted animal and mouse experiments to validate their findings. Injecting the released inflammatory particles into mice resulted in weight loss, indicating the harmful effects of these particles. However, mice with inhibited or removed genes associated with cell death did not experience weight loss, further confirming the role of inflammasome-mediated cell death in disease progression.
The study, conducted in collaboration with Professor Daesik Kim from the Sungkyunkwan University School of Medicine, was published online on November 27 in Cellular & Molecular Immunology, a globally recognized immunology journal. This research was supported by the National Research Foundation of Korea (NRF) grant that was funded by the Korean government (MSIT), by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI) that was funded by the Ministry of Health & Welfare, Republic of Korea, by grants from the National Institute of Health, Republic of Korea, by the Institute for Basic Science (IBS), by a research fund from UNIST), by a grant from Yuhan Corporation (SL), by the National Research Foundation of Korea (NRF) and the Center for Women In Science, Engineering and Technology (WISET) grant that was funded by the Ministry of Science and ICT (MSIT) under the Program for Returners into R&D (to JL).
Journal Reference
SuHyeon Oh, Jihye Lee, Jueun Oh, et al., “Integrated NLRP3, AIM2, NLRC4, Pyrin inflammasome activation and assembly drive PANoptosis,” Cell. Mol. Immunol., (2023).












