A research team, affiliated with UNIST has unveiled a key factor involved in the DNA damage response (DDR), homologous recombination (HR) and DNA interstrand crosslink (ICL) repair. According to the research team, their findings are expected to establish an effective control environment for chromosome instability (CIN), a major factor in cancer evolution, and further help combat malignant tumors .
Published in the January 2023 issue of Nucleic Acids Research, this breakthrough has been jointly led by Professor Hongtae Kim and Professor Kyungjae Myung in the Department of Biological Sciences at UNIST, in collaboration with Professor Yonghwan Kim and his research team from Sookmyung Women’s University.
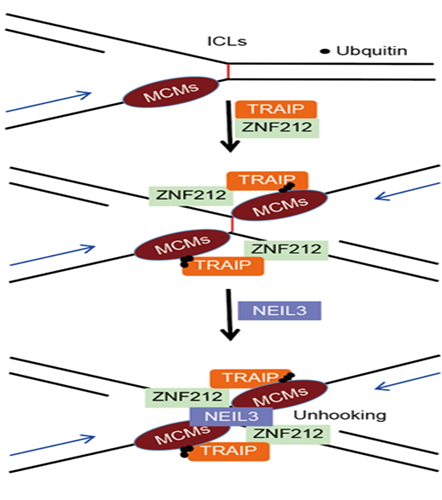
Figure 1. ZNF212 protein moves with TRAIP protein to the DNA binding damage site, promoting the recruitment of NEIL3 proteins to repair DNA damage and maintain the stability of the genome.
In this study, the research team demonstrated that, as a novel interaction partner of TRAIP, ZNF212 plays important roles in DNA damage signaling and HR for cell survival and genome maintenance, and likely act upstream of both NEIL3 and FA pathways for ICL repair.
They, then, further identified that TRAIP appears to function as important factor for ICL repair as a regulatory factor upstream of both the NEIL3 and FA pathways in mESC lines. Their findings also revealed that TRAIP functions in ICL repair, as a master regulator.
“Our findings together with mESC lines used in this study will be informative to understand molecular basis of the ICL repair pathways in detail,” noted the research team.
This study has been supported by the National Research Foundation of Korea (NRF), the Institute of Basic Science (IBS), and the Korean government (MSIT). Their findings have been published in the online version of Nucleic Acids Research, an internationally renowned journal in the fields of biochemistry and molecular biology.
Journal Reference
Hee Jin Chung, Joo Rak Lee, Tae Moon Kim, et al., “ZNF212 promotes genomic integrity through direct interaction with TRAIP,” Nucleic Acids Research (2023).












