DNA is well known as the blueprint of life, necessary for an organism to facilitate living processes. DNA can be damaged by various factors such as radical metabolites, radiation, and some toxic chemicals. As DNA is a molecule consisting of two strands, either one or both of the strands can be damaged.
Single-strand break (SSB) occurs when one of the two strands of DNA is damaged or broken. These are relatively mild damages that can be easily repaired by specialized enzymes that can seal the break and restore the integrity of the DNA molecule. On the other hand, a double-strand break (DSB) refers to when both strands of DNA are damaged. These are considered the most severe type of DNA damage, capable of causing genetic mutations or cell death.
Cells maintain genome integrity by having various pathways to repair DSBs. Among the several mechanisms for repairing DSBs, homologous recombination repair (HR) is one such mechanism that is highly precise and error-free, as it uses the undamaged sister chromatid as a template to repair DSBs. On the other hand, DNA repaired by polymerase theta-mediated end-joining (TMEJ) can result in the loss of some genetic information and cause mutations. Therefore, it is crucial to choose the appropriate DSB repair process to maintain genome integrity.
But how do cells select the right repair process? And what kinds of proteins are involved in the selection process?
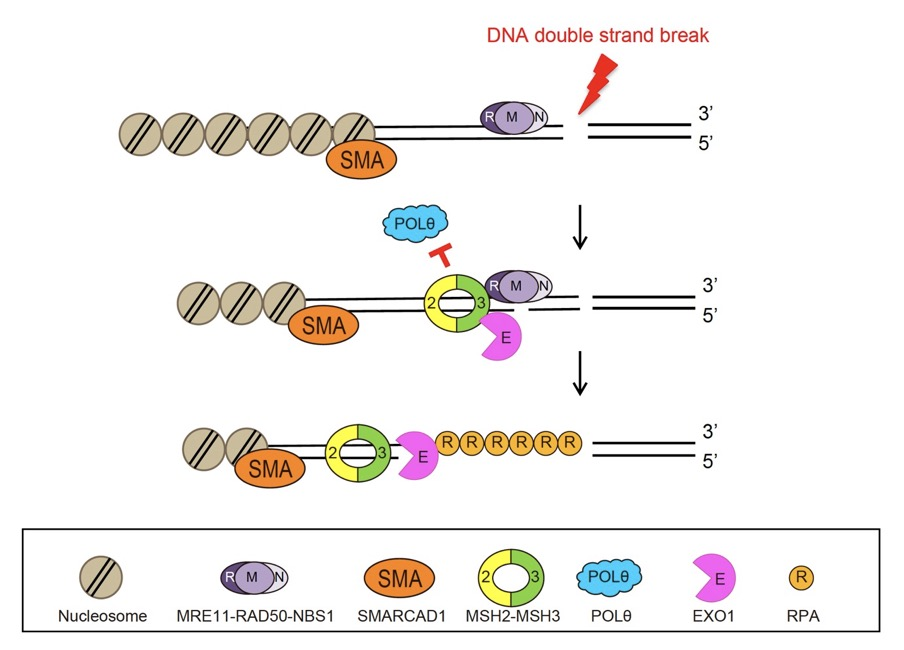
Figure 1. DSB repair by SMARCAD1, MSH2, and EXO1. At the early step of DNA double strand break repair, SMARCAD1 recruits MSH2-MSH3 complex to inhibit POLθ-mediated end joining and promote long range end resection by EXO1 through protein-protein interaction.
Led by Distinguished Professor Kyungjae Myung, Director of the Center of Genomic Integrity (CGI) within the Institute for Basic Science (IBS) at UNIST, the research teams of Professor Ja Yil Lee (Department of Biological Science, UNIST), and Professor Jung-Min Oh at Pusan National University have discovered that repair proteins involved in DSB repair, mismatch repair, and TMEJ are closely related and interact with each other during DSB repair process.
There are various repair mechanisms in our cells, each tailored to the type of DNA damage. For instance, DSBs are repaired by DSB repair proteins, while improperly paired DNA bases are repaired by mismatch repair proteins. Until now, most researchers thought that a specific type of DNA damage is only repaired by its corresponding DNA repair mechanism.
However, this study revealed that repair proteins previously thought to be responsible for different repair mechanisms can interact with each other to recognize damaged sites and select an appropriate repair mechanism.
Specifically, it was revealed that MSH2-MSH3, a DNA mismatch repair protein, actually plays a crucial role in the DSB repair process. The researchers observed the recruitment of fluorescent protein-labeled MSH2-MSH3 protein to the site of DSBs and revealed that this movement occurs through binding with the chromatin remodeling protein called SMARCAD1. The binding of MSH2-MSH3 to DSBs facilitates the recruitment of EXO1 (exonuclease 1) for long-range resection of damaged DNA.
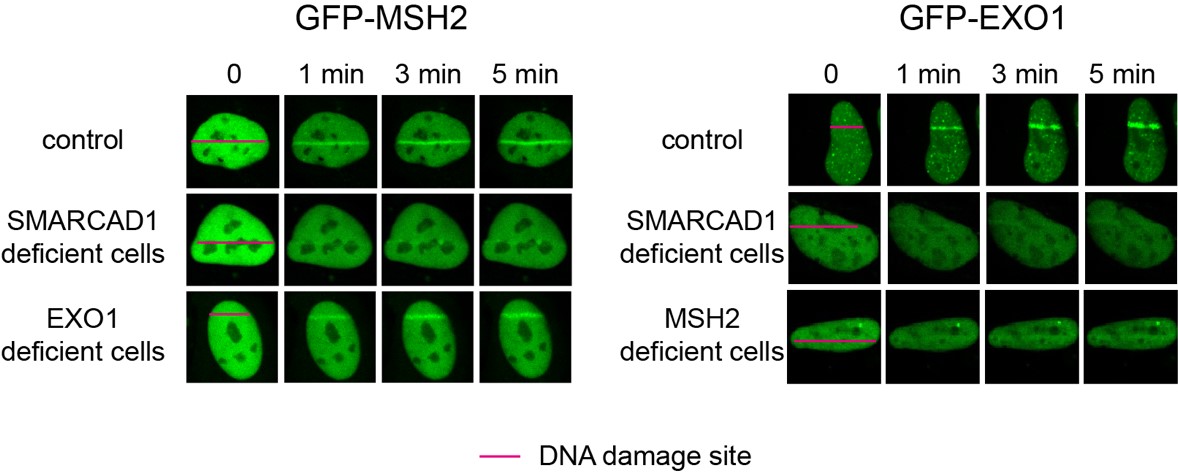
Figure 2. Laser microirradiation is used to mimic the DSBs in cells and measure the recruitment of green flurecent protein-labeled MSH2 (GFP-MSH2) or EXO1 (GFP-EXO1) to the DNA damage site (indicated by the red line) under a confocal microscope. The recruitment of GFP-MSH2 was inhibited in SMARCAD1-deficient cells, but not in EXO1-deficient cells. The recruitment of GFP-EXO1 was inhibited in SMARCAD1- or MSH2- deficient cells. These data indicates that MSH2 recruitment to DSBs depends on SMARCAD1, and then EXO1 is recruited.
After the long-range resection, the damaged DNA is repaired through error-free HR. Furthermore, it was discovered that the binding of MSH2-MSH3 inhibits the access of POLθ, which mediates a more error-prone TMEJ pathway, thereby preventing mutations that may occur during DSB repair.
“This research has revealed a new function of the mismatch repair protein MSH2-MSH3 in regulating DSB repair,” said Professor Myung. “The repair proteins that have been believed so far to act independently in the mismatch repair, double-stranded break repair, and TMEJ repair pathways are now shown to closely interact each other for proper maintenance of genomic integrity.”
Their findings have been published in the May 2023 issue of Nucleic Acids Research.
Kyungjae Myung
Distinguished Professor, Department of Biomedical Engineering, UNIST
IBS Center for Genomics Integrity
William Suh
IBS Communications Team
T: +82-42-878-8137
E:willisuh@ibs.re.kr
Story Source
Materials provided by Institute of Basic Science.
Notes for Editors
This press release is made available courtesy of the IBS Communications Team. The online version of the original article can be found HERE.
Journal Reference
Jung-Min Oh, Yujin Kang, Jumi Park, et al., “MSH2-MSH3 promotes DNA end resection during homologous recombination and blocks polymerase theta-mediated end-joining through interaction with SMARCAD1 and EXO1,” Nucleic Acids Res., (2023).