A team of researchers, led by Professor Yoon-Kyoung Cho of Life Science at UNIST has recently developed a new technique that effectively identifies cancer-causing substances in the urine or blood.
In the study, Professor Yoon-Kyoung Cho of Life Science, a group leader at IBS Research Center for Soft and Living Matter (CSLM) presented an integrated centrifugal microfluidic
platform (Exodisc), a device that isolates extracellular vesicles (EVs) from urine.
The results of the study has been published in the February issue of ACS Nano journal. The research team expects that this could be potentially useful in clinical settings to test urinary EV-based biomarkers for cancer diagnostics.

The Exodisc, developed by Professor Yoon-Kyoung Cho (School of Life Sciences)’s research team can effectively isolate cancer-causing substances in the urine or blood.
Extracellular vesicles (EVs) are cell-derived nanovesicles (40–1000 nm in size), present in almost all types of body fluids, which play a vital role in intercellular communication and are involved in the transport of biological signals for regulating diverse cellular functions.
Despite the increasing clinical importance of EVs as potential biomarkers in the diagnosis and prognosis of various diseases, current methods of EV isolation and analysis suffer from complicated procedures with long processing times. For instance, even ultracentrifugation (UC), the most commonly used method for EV isolation, requires time-consuming steps involving centrifugation and acquisition of large sample volumes, and the results suffer from low yield and purity.
To overcome these limitations, Professor Cho presented a new lab-on-a-disc platform for rapid, size-selective, and efficient isolation and analysis of nanoscale EVs from raw biological samples, such as cell-culture supernatant (CCS) or cancer-patient urine.
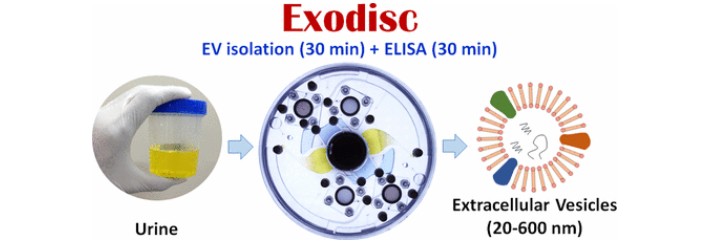
“The Exodisc is compoased of two independent filteration units (20nm and 600nm in size) within a disk-shaped chip to enable the processing of two different samples simulateously,” says Hyun-Kyung Woo (Combined M.S./Ph.D. student of Biomedical Engineering), the first author of the study. “Upon spinning the disc, the urine sample is transferred through two integrated nanofilters, allowing for the enrichment of unirary EVs within the size range of 20 to 600 nm.”
The hole sizes in nanoporous filters mounted on the Exodisc were set arbitrarily to seperate the nanoscale vesicles effectively from germs or unnecessary proteins. The direct detection of urinary EVs from patients with bladder cancer was validated successfully, using on-disc enzyme-linked immunosorbent assay (ELISA).
“Using Exodisc, it is possible to isolate EVs from raw samples within 30 minutes,” says Professor Cho. “The process of passing the filter through centrifugal force is automatically carried out, effectively recovering the enriched EVs.”
In the study, the research team examined clinical samples by analyzing urinary EVs from bladder cancer patients, using a tabletop-sized centrifugal microfluidic system. As a result, fully automated enrichment of unirary EVs in the size range of 20−600 nm was achieved within 30 min Quantitative tests.
“On-disc ELISA using urinary EVs isolated from bladder cancer patients showed high levels of CD9 and CD81 expression, suggesting that this method could be potentially useful in clinical settings to test urinary EV-based biomarkers for cancer diagnostics,” says Vijaya Sunkara of Life Sciences, the co-first author.
“We are currently conducting further studies to determine various diseases, including cancer by analyzing the collected nanoparticles,” says Professor Cho. “We hope this device can contribute significantly to the advancement of studies related to tumor biology and acceleration of the practical use of EV-based liquid biopsies for personalized medicine in clinical settings,” says Professor Cho.
The study has been conducted in collaboration with Professor Yoon-Keun Kim of the Institute of MD healthcare. It has been supported by the Korean Ministry of Health & Welfare, the Institute for Basic Science (IBS), and SRC.
Journal Reference
“Exodisc for Rapid, Size-selective, and Efficient Isolation and Analysis of Nanoscale Extracellular Vesicles from Biological Samples,” ACS Nano, (2017).

















