A research team, affiliated with UNIST has drawn attention for developing a novel and cost-effective technology for producing single-crystalline lithium-and-manganese-rich (Li-/Mn-rich) and Ni-rich layered cathodes, which are key to increasing the electric vehicle range by more than 30%.
This breakthrough has been carried out by Distinguished Professor Jaephil Cho in the School of Energy and Chemical Engineering at UNIST, in collaboration with Professor Ju Li from MIT in the United States. In this study, the research team invented a new mechanochemical activation process that offers a general solution to the conundrum of synthesizing coarse single-crystal cathodes with Li-/Mn-rich or Ni-rich chemistry, which differs from the equipment- and energy-intense and long-duration mechanochemical routes that are difficult to scale up. Their approach was based on interfacial reactive wetting, mediated by transient eutectic salts in situ melted by moderate mechanical agitations, to form a colloidal suspension of nanosized oxides dispersed in liquified lithium salts.
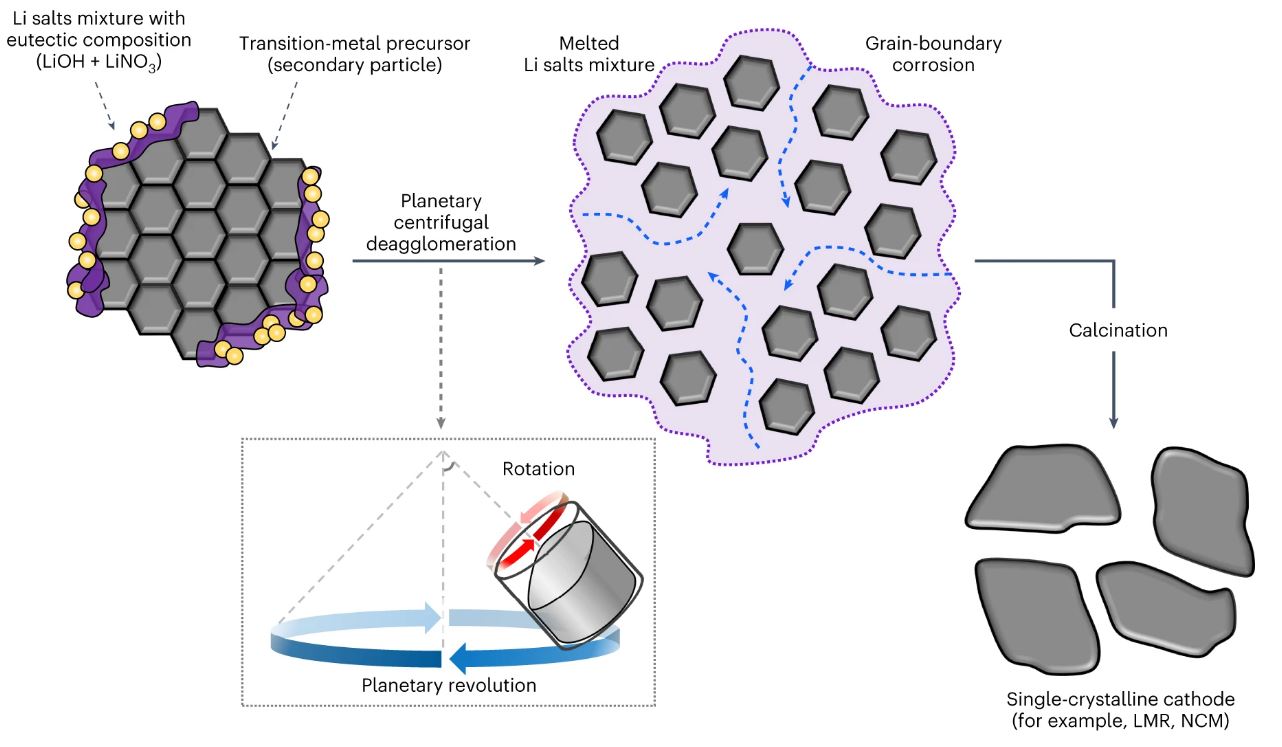
Figure 1. Planetary centrifugal deagglomeration for single-crystalline cathode synthesis. In the process, the transient molten Li salts (purple) corrode the grain boundaries (blue dashed arrows) of the transition-metal-oxide precursor (grey hexagons) and separate approximately ten micron-sized secondary particles into nano-sized primary ones. The molten salts, that is, a mixture of LiOH and LiNO3 at/near the eutectic composition, serve as a corrosive liquid during the planetary centrifugal deagglomeration process and the Li source during the calcination process of layered cathode synthesis. Successful examples include Li-/Mn-rich layered cathode LMR and Ni-rich layered cathode NCM.
In this study, the research team combined lithium nitrate, lithium hydroxide, and polycrystalline transition metal precursors dissolved by eutectic composition at a certain ratio. It was mixed for 12 minutes at a rate of 2000 times/min using a pneumatic and electronic mixer. It has been shown that liquid lithium salt-transition metal nanoparticle complex can be created as heat-melted powders generated from contact penetrate into the interface of polycrystalline particles (crystalline grain erosion). The research team developed a technology that heats the complex at less than 800 degrees for 10 hours to form a fully crystallized single crystal with a size of several micro.
This technology can be applied not only to nickel-rich anodes but also to lithium-manganese-rich anodes. Lithium-manganese rich cathodes are materials that contain high Mn content (60% or more). In addition, since the content of lithium is higher than the content of the transition metal, it is a material that exhibits a high capacity of 250 mAh/g or higher at a high voltage of 4.5 V or higher. As the Mn content increases, the thermal treatment temperature required for synthesis increases. In particular, when the Mn content is 60% or more, the synthesis is not possible as a single crystal even if the Mn content is heated at 900°C or more for 12 hours or more. However, through this study, it was announced that it can be synthesized into single crystalline particles of 1 micrometer size even if the Mn content is more than 60%.
The research team used this study to measure battery performance. A single crystal cathode material having the same composition as a polycrystalline material (NCM811) synthesized through a general process was evaluated on a lithium metal battery. This single crystal cathode material showed performance equivalent to 92% of the existing capacity even after 200 times of charging and discharging, and showed a life retention rate improved by about 12% compared to a polycrystalline material of the same composition. In addition, gas generation and resistance growth rates have improved by more than 30%, which is expected to play a major role in improving safety, a chronic problem of electric vehicles.
The findings of this study have been published in the March 2023 issue of Nature Energy, an internationally-renowned journal in the area of energy.
Journal Reference
Moonsu Yoon, Yanhao Dong, Yimeng Huang, et al., “Eutectic salt-assisted planetary centrifugal deagglomeration for single-crystalline cathode synthesis,” Nature Energy, (2023).