A breakthrough study, jointly led by Professor Jang Hyun Choi and Professor Sung Ho Park from the Department of Biological Sciences at UNIST has identified an important factor involved in the development of non-alcoholic fatty liver disease (NAFLD) caused by obesity. The research team discovered that Thrap3, a protein associated with thyroid hormone receptors, plays a significant role in exacerbating NAFLD by inhibiting the activity of adenosine monophosphate-activated protein kinase (AMPK), a key regulator of fat metabolism in the liver.
NAFLD encompasses various metabolic diseases such as fatty hepatitis and cirrhosis resulting from excessive fat accumulation. Despite its prevalence, effective treatments for NAFLD have been limited. However, this groundbreaking research sheds light on potential therapeutic approaches.
Through animal experiments conducted on rats, the research team demonstrated that Thrap3 directly binds to AMPK within the liver. This interaction prevents AMPK from translocating from the nucleus to the cytoplasm and impairs autophagy—a process crucial for breaking down triglycerides and reducing cholesterol levels. In essence, inhibiting Thrap3 expression presents a promising avenue for effectively treating NAFLD.
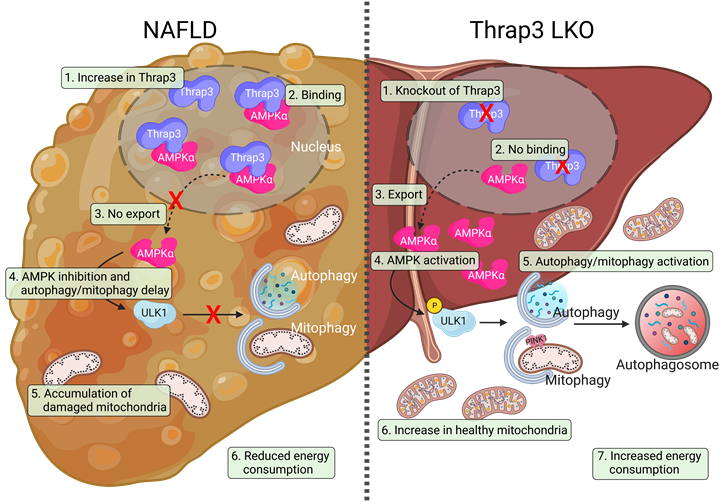
Figure 1. Schematic diagram of the mechanism by which Thrap3 affects NAFLD through translocation of AMPK. Dysregulation of autophagy/mitophagy contributes to the progression of nonalcoholic fatty liver disease (NAFLD). In NAFLD, increased Thrap3 suppresses autophagy/mitophagy by sequestering AMPK in the nucleus. Inhibition of mitophagy does not effectively remove damaged mitochondria and maintain healthy mitochondria, and as a result, it leads to a decrease in energy consumption and exacerbates NAFLD progression. Liver-specific Thrap3 knockout mice show improved lipid accumulation, metabolic parameters, and mitochondrial function and enhanced autophagy/mitophagy in the NAFLD model. Thrap3 may be a potential therapeutic target for preventing and treating NAFLD by regulating the AMPK/autophagy/mitophagy axis.
“We have encountered significant challenges while developing treatment strategies for non-alcoholic fatty liver disease. However, our discovery of the Thrap3 gene provides us with an effective method to tackle this condition,” commented Professor Choi.
Additionally, it was confirmed that suppressing Thrap3 expression effectively improves non-alcoholic steatohepatitis—an inflammatory disease stemming from fatty liver.
Supported by funding from Korea Research Foundation under the Ministry of Science and ICT, National Mouse Phenotype Analysis Group (KMPC), and UNIST Future Lead Project; this research was published online on August 1st in Experimental and Molecular Medicine. Professor Jang Hyun Choi, alongside Professor Sung Ho Park, served as corresponding authors of the paper, with Dr. Hyun-Jun Jang and Dr. Yo Han Lee from the Department of Biological Sciences at UNIST participating as co-authors.
Journal Reference
Hyun-Jun Jang, Yo Han Lee, Tam Dao, et al., “Thrap3 promotes nonalcoholic fatty liver disease by suppressing AMPK-mediated autophagy,” Exp. Mol. Med., (2023).